
FDA Draft Guidance - PCCP Recommendations AI ML
5
$ 15.99
In stock
(167)
Product Description

Sierra Labs Blog

Expert: FDA's Highly Anticipated PCCP Guidance Isn't Anything Too 'Surprising' :: Medtech Insight

FDA drafts AI-enabled medical device life cycle plan guidance

AI developers should build robust change control protocols despite absence of FDA guidance

FDA Release New Guidance for Modifications to AI/ML-Enabled Devices - Oxford Global
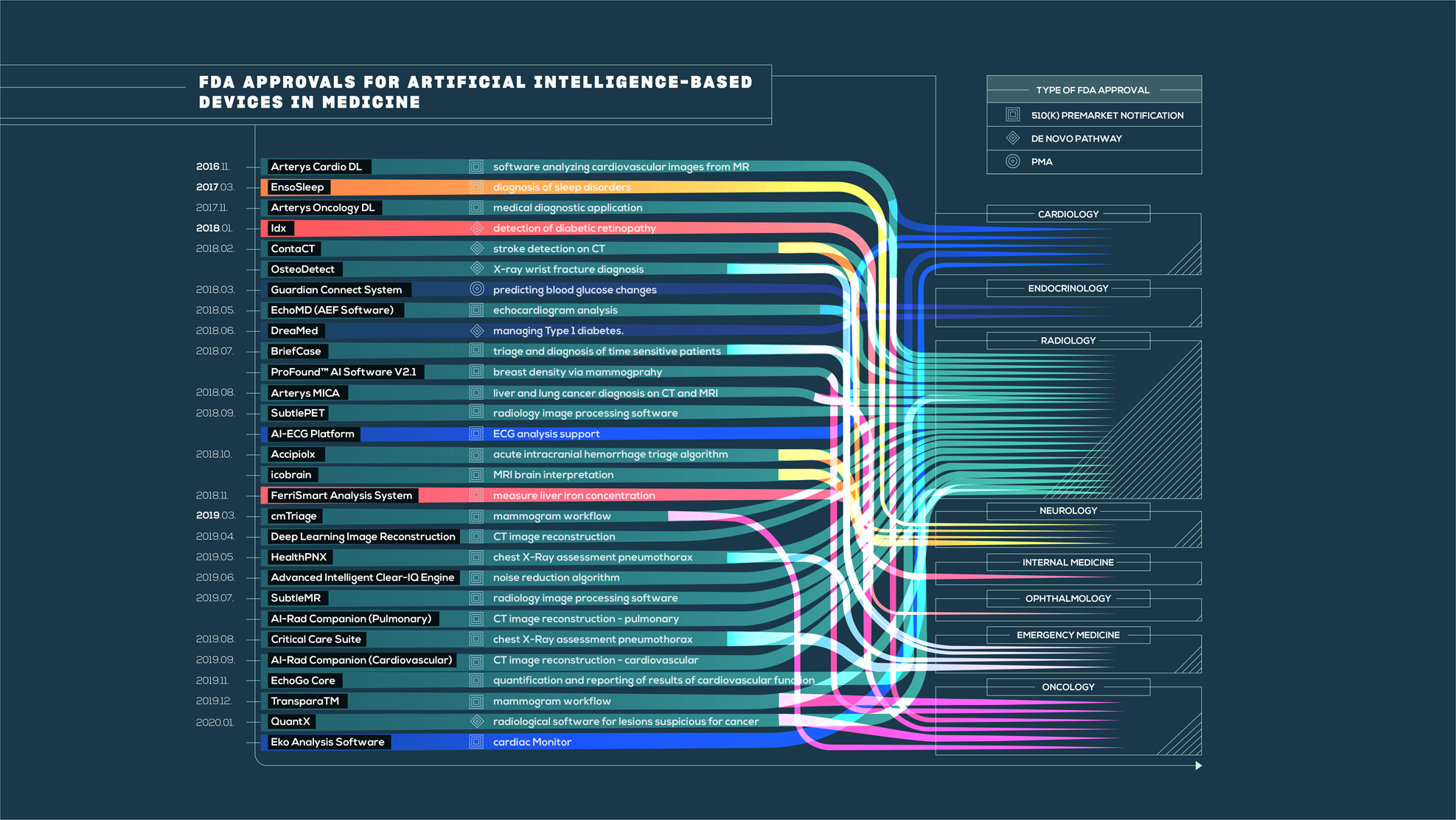
The state of artificial intelligence-based FDA-approved medical devices and algorithms: an online database

FDA Predetermined Change Control Plan (PCCP) Guidance for Artificial Intelligence (AI)

New FDA Draft Guidance on Predetermined Change Control Plans for AI/ML-Enabled Devices – CENIT Consulting
.png)
FDA Guidance on Artificial Intelligence (AI) in Medical Devices

US FDA Pre-Market Notification - 510(k)

FDA Publishes Draft Guidance for Predetermined Change Control Plan for AI/ML-Enabled Device Software Functions

Know the basics about Meeting with the FDA for Medical Device Pre-Submissions.

Technology Roundup – Augmented Intelligence, Cybersecurity, Augmented Reality — Agilis Consulting Group

FDA Guidance on Artificial Intelligence (AI) in Medical Devices






:format(jpeg)/cdn.vox-cdn.com/uploads/chorus_image/image/48638895/usa-today-8913414.0.jpg)





